Trastuzumab Deruxtecan Demonstrates Clinical Activity Across Numerous HER2+ Solid Tumors
In the phase 2 DESTINY-PanTumor02 trial, trastuzumab deruxtecan elicited clinical activity across a range of HER2 expressing solid tumors.
Funda Meric-Bernstam, MD

Fam-trastuzumab deruxtecan-nxki (Enhertu) induced responses among patients with a wide range of HER2-positive solid tumors, according to findings from the phase 2 DESTINY-PanTumor02 trial (NCT04482309) presented during the 2023 ASCO Annual Meeting.
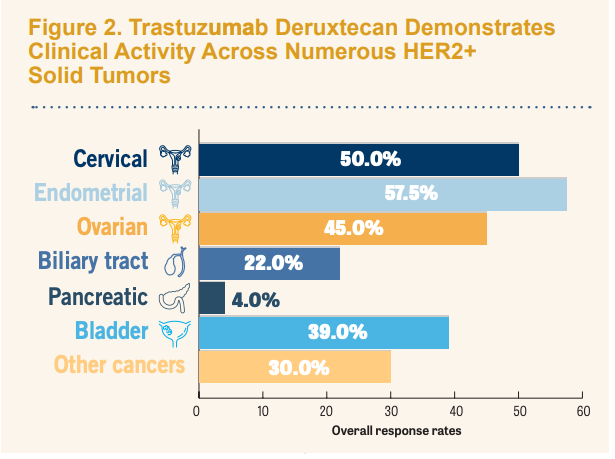
Results from the study showed that at the November 16, 2022, data cutoff, patients with cervical (n = 40), endometrial (n = 40), ovarian (n = 40), biliary tract (n = 41) pancreatic (n = 25), bladder (n = 41), and other cancers (n = 40) experienced investigator-assessed overall response rates (ORRs) of 50.0%, 57.5%, 45.0%, 22.0%, 4.0%, 39.0%, and 30.0%, respectively. The complete response (CR) rates were 5.0%, 17.5%, 10.0%, 2.4%, 0%, 2.4%, and 0% respectively. The ORR in the total population (N = 267) was 37.1%, including a 5.6% CR rate.
“Trastuzumab deruxtecan is a potent antibody-drug conjugate that has become a standard of care for HER2-low as well as HER2-positive breast cancer, HER2-positive gastric cancer, and HER2-mutant lung cancer,” Funda Meric-Bernstam, MD, said in a presentation of the data. Meric-Bernstam is chair of the Department of Investigational Cancer Therapeutics—the Phase I Program, medical director of the Institute for Personalized Cancer Therapy (IPCT), and the Nellie B. Connally Chair in Breast Cancer at MD Anderson Cancer Center in Houston, Texas. “However, HER2 is expressed across a variety of other tumor types, and for these patients there are no approved HER2-targeted treatments, and often the disease is refractive to standard-of-care therapies. In early-phase studies, trastuzumab deruxtecan demonstrated activity in several tumor types, including salivary gland, biliary [tract], and endometrial cancers.”
DESTINY-PanTumor02 was an open-label, multicenter study of patients with advanced solid tumors who were not eligible for curative therapy. Patients needed to be in their second line or later of therapy, have a HER2 expression by immunohistochemistry (IHC) of 3+ or 2+, and an ECOG performance status of 1 or 0. Prior HER2-directed therapy was permitted. Trastuzumab deruxtecan was administered at a dose of 5.4 mg/kg every 3 weeks in all cohorts.
The primary end point was confirmed ORR by investigator. Secondary end points included disease control rate (DCR), duration of response (DOR), progression-free survival (PFS), overall survival (OS), and safety.
The safety profile of trastuzumab deruxtecan was found to be consistent with the known profile of the agent. In the overall population, common any-grade treatment-emergent adverse events (TEAEs) included nausea (54.3%), fatigue (37.8%), neutropenia (32.6%), anemia (25.8%), diarrhea (25.5%), vomiting (24.3%), decreased appetite (16.5%), thrombocytopenia (16.5%), alopecia (16.1%), and leukopenia (10.1%). These AEs occurred at a grade 3 or higher severity in 3.4%, 6.0%, 19.1%, 8.6%, 3.7%, 1.5%, 1.5%, 5.2%, 0.0%, and 2.6% of patients, respectively.
Regarding interstitial lung disease and pneumonitis related to treatment with trastuzumab deruxtecan, any-grade events were reported in 7.5% of patients. Most instances were grade 2 (4.5%), although grade 1 (2.2%), grade 3 (0.4%), and grade 5 (0.4%) events were also reported.
Additional findings from the study revealed that the median DORs were 9.8 months (95% CI, 4.2-not evaluable [NE]), not reached (NR; 95% CI, 9.9-NE), 11.3 months (95% CI, 4.1-NE), 8.6 months (95% CI, 2.1-NE), not reached, 8.7 months (95% CI, 4.3-11.8), and not reached (95% CI, 4.1-NE) in the cervical, endometrial, ovarian, biliary tract, pancreatic, bladder, and other cohorts, respectively. The 12-week DCR rates were 67.5%, 80.0%, 70.0%, 65.9%, 36.0%, 70.7%, and 75.0%, respectively.
Notably, patients with a HER2 status of IHC 3+ generally experienced heightened response rates. The ORRs among patients with an IHC of 3+ were 75.0%, 84.6%, 63.6%, 56.3%, 0.0%, 56.3%, and 44.4%, in the cervical (n = 8), endometrial (n = 13), ovarian (n = 11), biliary tract (n = 16), pancreatic (n = 2), bladder (n = 16), and other (n = 9) cohorts, respectively. Comparatively, the ORRs for patients with a HER2 of IHC 2+ were 40.0%, 47.1%, 36.8%, 0.0%, 5.3%, 35.0%, and 18.8 % in the cervical (n =20), endometrial (n = 17), ovarian (n = 19), biliary tract (n = 14), pancreatic (n = 19), bladder (n = 20), and other (n = 16) cohorts, respectively.
The median DOR for all patients with an IHC of 3+ was 22.1 months (95% CI, 9.3-NE) and was 9.8 months (95% CI, 4.2-12.6) among those with an IHC of 2+.
“These were durable responses,” Meric-Bernstam said. “The trial is still ongoing, and we will be reporting OS and PFS [data] at a later date. However, data to date suggest that trastuzumab deruxtecan has broad activity and could represent a new therapy option in patients that are HER2-positive.”
Reference
Meric-Bernstam F, Makker V, Oaknin A, et al. Efficacy and safety of trastuzumab deruxtecan (T-DXd) in patients (pts) with HER2-expressing solid tumors: DESTINY-PanTumor02 (DP-02) interim results. J Clin Oncol. 2023;41(suppl 17):LBA3000.



